Scientist of the Day - James Franck
On Apr. 24, 1914, James Franck, a 32-year-0ld German physicist, read a paper to the Physical Society of Berlin, reporting the results of experiments performed by himself and his young assistant, Gustav Hertz. The Franck-Hertz experiment described in that paper is now one of the classic and transformative experiments in modern physics, right up there with the Davidson-Germer experiment (1927), the Stern-Gerlach experiment (1922), the Millikan oil-drop experiment (1913), and the Michelson-Morley experiment (1889). It would earn Franck and Hertz a shared Nobel Prize in 1925.
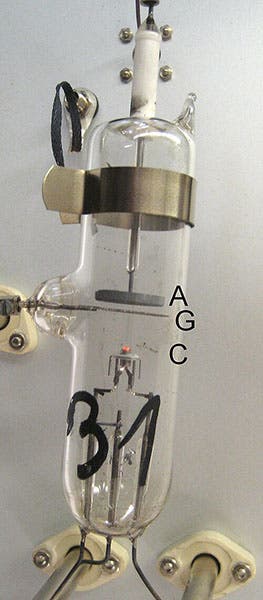
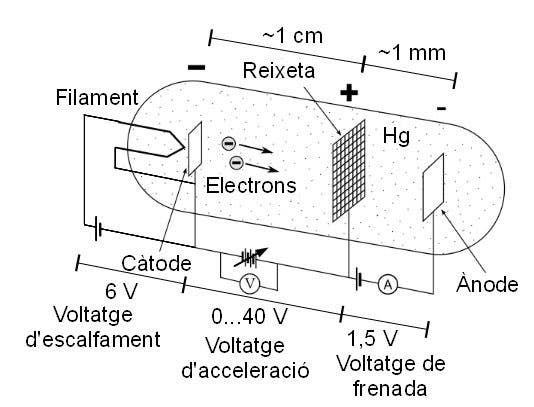
The experiment was simplicity itself. In a triode, a vacuum tube with three elements, they put some mercury, which vaporized. They heated the cathode, so that it would emit electrons, which would be attracted to the anode. A wire-mesh grid between cathode and anode was connected to an adjustable voltage source, and controlled the speed of the electrons.

First paragraph of paper announcing the results of the Franck-Hertz experiment, "Über Zusammenstöße zwischen Elektronen und Molekülen des Quecksilberdampfes und die Ionisierungsspannung desselben" (On the collisions between electrons and molecules of mercury vapor and the ionization potential of the same), by James Franck and Gustav Hertz, Verhandlungen der Deutschen Physikalischen Gesellschaft, vol. 16, p. 457, 1914 (Linda Hall Library)
Franck and Hertz were trying to determine how electrons interact with the atoms in the mercury vapor. They discovered that the current in the tube increased until the electrons had an energy of 4.9 eV (electron-volts), when suddenly the current dropped to near zero. The electrons were now interacting with the mercury atoms, either knocking their electrons into a higher orbit, or causing them to emit light. At 4.8 eV, the cathode electrons either bounced off the mercury atoms or passed by; with 4.9 eV, they gave up all their energy, all at once. What was going on?
Unbeknownst to Franck, the Danish physicist, Niels Bohr, the year before, had proposed that the energy of the electrons in a hydrogen atom is quantized. There is a minimum amount of energy that an electron can have, and that is the energy of the lowest orbit in the atom. Give another quantum of energy to the electron, and it will leap (a quantum leap) to the second orbital level in the atom. Leave it alone, and it will emit a photon of light with a very specific wavelength, and drop down to the lower orbit. Bohr was able to explain all four of the wavelengths emitted by the hydrogen atom with his quantum theory of the hydrogen atom
Franck and Hertz did not know it in 1914, but their experiment could best be explained – indeed, only be explained – by Bohr’s quantum theory. An electron with less than 4.9 eV of energy was unable to interact with anything. Once it reached that energy, it could knock an electron of mercury into a higher orbit. The light emitted by the stimulated atoms was always 254 nanometers, with an energy exactly equaling 4.9 eV. The Franck-Hertz experiment was the first independent confirmation of the Bohr atom. It was Bohr, not Franck, who first realized this, but by the end of WWI, which fully occupied Franck for 4 years, Franck was in full agreement with Bohr's interpretation. And Albert Einstein was full of praise for their elegant experiment.
We told this story six years ago, and told it differently, in our first post on Franck, during the early months of the COVID era, when we worked from home and were restricted, for some reason, to two images. We refer you to that post for more details on Franck's life and later career – and for the two images we were allowed, one a photo with Einstein and Bohr. Here we offer all the images we couldn't show the first time, including some diagrams and photos of the experiment, and various group photos, and the official Nobel photo.
Our fifth image shows Franck with Max Born, the quantum physicist who brought Franck to the University of Göttingen in 1920 and who was a best friend of not only Franck, but of Einstein. The last photo, of the Göttingen physics staff in 1927, provides a rare glimpse of Hertha Sponer, one of the few women physicists in Germany, who would emigrate to the United States and eventually marry Franck, after his first wife died. A post on Sponer is in order.
William B. Ashworth, Jr., Consultant for the History of Science, Linda Hall Library and Associate Professor emeritus, Department of History, University of Missouri-Kansas City. Comments or corrections are welcome; please direct to ashworthw@umkc.edu.