Scientist of the Day - Stanley Miller
Stanley Miller, an American biochemist, was born Mar. 7, 1930. In 1951, Miller was a new graduate student at the University of Chicago, when he attended a colloquium given by Harold Urey, a Nobel prize winner for his discovery of deuterium, in which Urey lamented the lack of effort being made to test the idea that a “primordial soup” on the early earth had given rise to life, an idea that had been advanced by J.B.S. Haldane and Aleksandr Oparin in the 1920s. Since Miller was looking for an interesting project for his PhD, he opted to follow up on Urey's challenge, and asked Urey to be his advisor. Urey at first refused, for good reason, since prospective PhD students who pursue experiments that fail do not get PhDs, and Urey was not at all sure that the proposed experiment would work. But Miller was persuasive, Urey agreed to mentor it, and Miller, in the fall of 1952, set up a laboratory experiment in which he placed methane, ammonia, carbon dioxide, and water vapor in a flask, gases which were surmised to have made up the primordial atmosphere of the Earth.
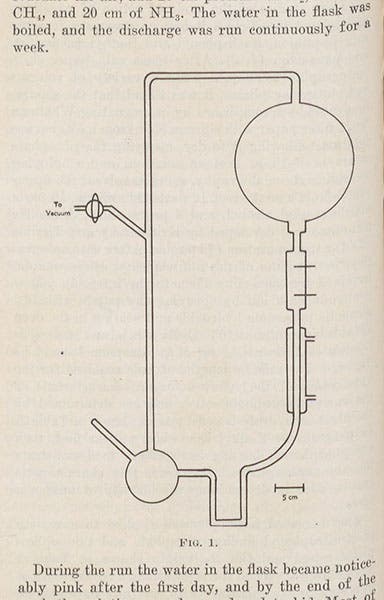
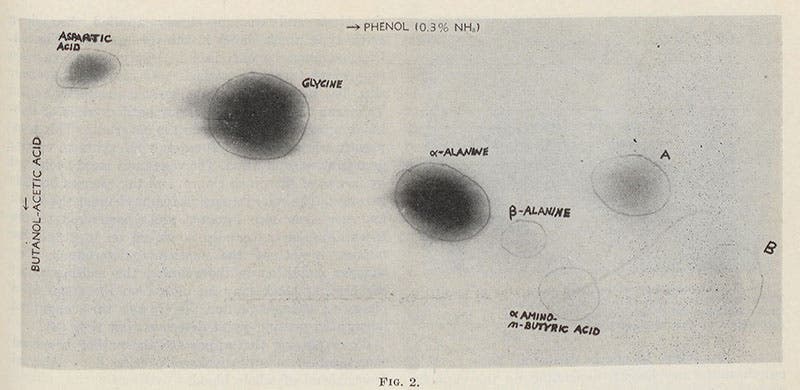
The set-up could not have been simpler – a bulbous glass vessel containing the gases, connected by tubing to other vessels with electrodes to produce sparks, a heat source, a cooling condenser, and a collecting flask to hold any molecules that might be produced. Miller found, when he tested the collecting solution after some time, that it contained several amino acids, the organic molecules that are the building blocks of proteins. That was a very exciting result. Miller wrote up a paper and, early in 1953, sent it off to Science, the premier scientific journal in the U.S. When he had not heard back after a month, he told Urey. Urey called up the editor and demanded the paper back, saying they would find a more responsive journal for it; the editor then immediately contacted Miller and said that the paper had been accepted. Believe me, you do not want to try this unless you have a Nobel Prize in your pocket – editors of science journals ordinarily do not take guff from anyone, except the laurelled few, like Urey.
And so the paper, “A production of amino acids under possible primitive Earth conditions," with Stanley Miller as sole author, was published in Science on May 15, 1953, and you can see the entire paper here – all two pages of it, plus details of the opening paragraph (fourth image), the apparatus (second image), and a photograph of the paper chromatogram that revealed the presence of glycine and alanine, two amino acids (sixth image). It right away became one of the classic papers in the history of the life sciences, showing that it was possible for the organic molecules that make up living things to form naturally under primitive earth-like conditions. It has been repeated thousands of times, and even though we now know that the primordial gases on Earth 4 billion years ago were not those suggested to Miller for his experiment, it doesn’t appear to make any difference – seemingly no matter what you put in the flask, organic molecules emerge from the collector.
One of the best features of the story is that Urey refused to have his name added as coauthor (which was customary for a PhD supervisor), saying that if his name were on the paper, no one would give Miller any credit. As it was, Miller got a great deal of credit, and he became as famous as any graduate student is likely to become, especially when the popular press found out that Miller had made “life in a test tube” (which of course he hadn’t). So it seems only fair that, in spite of Urey’s disavowals, Miller’s experiment is often referred to as the “Miller-Urey experiment” (indeed, this happens to be the title of the Wikipedia article on the subject). It is certain that without Urey and his help, there would have been no experiment at all.
Miller went on to spend most of his career at the University of California, San Diego (where Urey also moved when Chicago “retired” him), continuing to investigate the biochemistry of the early Earth. But Miller must have found it wearisome that whenever he was asked to participate in a photo session, he was always requested to pose with the apparatus from his 1952 experiment. Indeed, it is hard to find a portrait of Miller in which the familiar circular set-up of glass vessels and tubing are absent; see, for example, the one we include here (first image), which must haven been taken in the 1980s, 30 years after young Miller was gamely pursuing his PhD. I suppose there are worse things than becoming famous as a young person for a truly classic piece of scientific investigation. But I am sure that Miller must have thought, on some of these occasions, wouldn’t you like a picture of me with my dog?
Dr. William B. Ashworth, Jr., Consultant for the History of Science, Linda Hall Library and Associate Professor emeritus, Department of History, University of Missouri-Kansas City. Comments or corrections are welcome; please direct to ashworthw@umkc.edu.