Scientist of the Day - James Prescot Joule
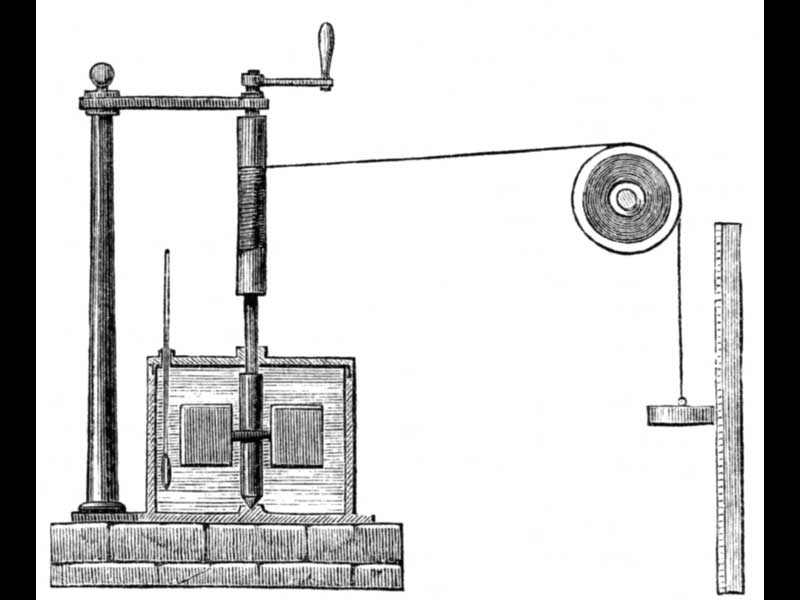
James Joule, an English physicist, was born Dec. 24, 1818. Joule was one of a dozen physicists who independently discovered the principle of the conservation of energy between 1843 and 1847. Joule did so by demonstrating what he called the mechanical equivalent of heat. He inserted a small paddle wheel on a vertical axis into a container of water, wrapped a long cord around the axle, tied a weight to the other end of the cord, ran it over a pulley, and then allowed the weight to fall, turning the paddles, and agitating and heating up the water. On taking the temperature of the water before and after, Joule found a direct correlation between the distance the weight fell and the increase in temperature of the water. Motion was being converted into heat at a predictable rate, and the measure of both heat and motion was a new entity, called energy. In a surprisingly rational move by those who supervise such things, the basic unit of energy, the joule, was named after our honoree. Joule’s original calorimeter is in the Science Museum, London (see second image above); a diagram helps visualize how it worked (third image above).
Joule’s former home in Manchester is now an Energy Centre for the University of Salford (fourth image above) and carries an informative blue plaque (fifth image). There is a bust of Joule by John Cassidy in Worthington Park, Sale, Cheshire (first image).
Dr. William B. Ashworth, Jr., Consultant for the History of Science, Linda Hall Library and Associate Professor, Department of History, University of Missouri-Kansas City